Here are some important questions that you might encounter in a Class 10 Science exam:
What Are the Three States of Matter and What Are Their Characteristics?

The three states of matter are solid, liquid, and gas.
- Solids have a fixed shape and a fixed volume. They are not easily compressible and have a relatively high density.
- Liquids have a fixed volume but take the shape of their container. They are not easily compressible and have a lower density than solids.
- Gases have neither a fixed shape nor a fixed volume. They are highly compressible and have a very low density.
The characteristics of the three states of matter are determined by the arrangement and movement of the particles (atoms, molecules, or ions) that make up the substance. Solids have particles that are closely packed together and are not free to move around, while the particles in liquids are more free to move but are still relatively close together. In gases, the particles are widely spaced and are free to move in any direction.
How Does the Concentration of a Solution Affect Its Properties?
The concentration of a solution refers to the amount of solute (the substance being dissolved) in a given amount of solvent (the substance doing the dissolving). The concentration of a solution can affect its properties in several ways:
- Density: Generally, the more concentrated a solution is, the denser it will be.
- Boiling point: Increasing the concentration of a solution will raise its boiling point. This is because the solvent molecules have to overcome a greater number of solute molecules in order to escape into the vapor phase.
- Freezing point: Increasing the concentration of a solution will also lower its freezing point. This is because the solute molecules interfere with the movement of the solvent molecules, making it more difficult for the solution to freeze.
- Osmotic pressure: The concentration of a solution can affect its osmotic pressure, which is the pressure that would need to be applied to a solution to stop osmosis (the movement of solvent molecules through a membrane into a more concentrated solution).
- Solubility: The solubility of a solute (the amount of solute that can dissolve in a given amount of solvent at a given temperature) can be affected by the concentration of the solution. In general, the solubility of a solute increases as the concentration of the solution increases.
Related Article: Human Body: Anatomy, facts and functions
What Is the Relationship Between Pressure and Volume in a Gas?
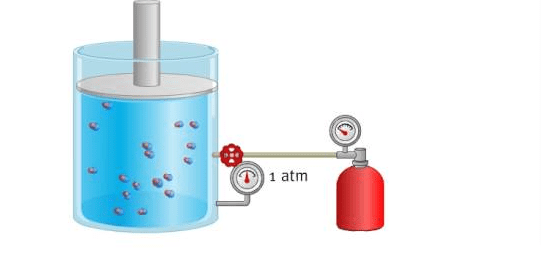
The relationship between pressure and volume in a gas is described by Boyle’s law, which states that, at a constant temperature, the pressure of a gas is inversely proportional to its volume. This means that if the pressure of a gas increases, its volume will decrease, and if the pressure decreases, its volume will increase.
For example, if you have a gas in a container with a fixed volume, and you increase the pressure on the gas (by squeezing the container, for example), the volume of the gas will decrease. Conversely, if you decrease the pressure on the gas (by expanding the container), the volume of the gas will increase.
This relationship between pressure and volume can be described by the equation PV = k, where P is the pressure of the gas, V is its volume, and k is a constant. This equation shows that the product of the pressure and volume of a gas is always constant, as long as the temperature remains the same.
What Is the Process of Photosynthesis and How Does It Benefit Plants?

Photosynthesis is the process by which plants, algae, and some bacteria convert light energy into chemical energy in the form of glucose (a sugar). This chemical energy is stored in the bonds of the glucose molecule and is used by the organism to power its metabolic processes.
Photosynthesis occurs in the chloroplasts of plant cells, and it involves two stages: the light-dependent reactions and the light-independent reactions (also known as the Calvin cycle).
The light-dependent reactions take place in the thylakoid membranes of the chloroplasts and involve the absorption of light by pigments such as chlorophyll. This absorbed light energy is used to split water molecules into hydrogen ions (H+) and oxygen atoms (O). The hydrogen ions are used to generate ATP (adenosine triphosphate), a molecule that stores chemical energy, and NADPH (nicotinamide adenine dinucleotide phosphate), a molecule that transfers reducing equivalents (electrons and hydrogen ions) to the light-independent reactions.
The light-independent reactions take place in the stroma of the chloroplasts and involve the fixation of carbon dioxide (CO2) into glucose using the energy from ATP and NADPH. This process is called the Calvin cycle.
Photosynthesis benefits plants in several ways. It provides them with a source of energy for their metabolic processes, and it also allows them to produce their own food. The oxygen produced during photosynthesis is released into the atmosphere as a byproduct, and this oxygen is essential for the survival of animals and other organisms that breathe oxygen. Additionally, the process of photosynthesis removes carbon dioxide from the atmosphere, which helps to mitigate the greenhouse effect and global warming.
How Do Living Organisms Maintain Homeostasis?
Homeostasis is the process by which living organisms maintain a stable internal environment in response to changing external conditions. This process is necessary for the organism to function properly and to survive.
Living organisms maintain homeostasis through a variety of mechanisms, including:
- Negative feedback: This is a common mechanism for maintaining homeostasis in which a change in a physiological variable triggers a response that counteracts the initial change. For example, if your body temperature starts to rise, your sweat glands will start to produce sweat, which will help to cool your body down and bring your temperature back to normal.
- Hormones: Hormones are chemical messengers that are produced by glands in the body and are released into the bloodstream. They help to regulate various functions in the body and maintain homeostasis by activating or inhibiting certain processes.
- Enzymes: Enzymes are proteins that catalyze chemical reactions in the body. They help to maintain homeostasis by regulating the rate of these reactions and ensuring that they occur at the appropriate times and in the appropriate amounts.
- Membranes: The cell membrane is a selectively permeable barrier that surrounds cells and helps to maintain homeostasis by controlling the movement of substances into and out of the cell.
Overall, the maintenance of homeostasis is essential for the survival and proper functioning of living organisms. It allows them to adapt to changing external conditions and maintain a stable internal environment.
What Are the Different Types of Chemical Reactions and How Can They Be Identified?

There are several types of chemical reactions, and they can be classified based on a variety of criteria, including the type of reactants, the type of products, and the energy changes that occur during the reaction. Some common types of chemical reactions include:
- Combination reactions: These reactions involve the combination of two or more reactants to form a single product. An example is the reaction between hydrogen and oxygen to form water: 2H2 + O2 -> 2H2O
- Decomposition reactions: These reactions involve the breakdown of a single reactant into two or more products. An example is the decomposition of water into hydrogen and oxygen gas when an electric current is passed through it: 2H2O -> 2H2 + O2
- Displacement reactions: These reactions involve the replacement of one element by another in a compound. An example is the reaction between zinc and copper(II) sulfate to form copper and zinc sulfate: Zn + CuSO4 -> Cu + ZnSO4
- Redox reactions: These reactions involve a transfer of electrons from one species to another. They can be identified by the presence of a reducing agent (a species that loses electrons) and an oxidizing agent (a species that gains electrons). An example is the reaction between zinc and hydrochloric acid to form zinc chloride and hydrogen gas: Zn + 2HCl -> ZnCl2 + H2
- Acid-base reactions: These reactions involve the transfer of a proton from an acid to a base. They can be identified by the presence of an acid (a species that donates a proton) and a base (a species that accepts a proton). An example is the reaction between hydrochloric acid and sodium hydroxide to form sodium chloride and water: HCl + NaOH -> NaCl + H2O
What Is the Structure of an Atom and How Does It Determine the Properties of an Element?
An atom is the basic unit of matter that represents an element. It is composed of three types of subatomic particles: protons, neutrons, and electrons.
The protons and neutrons are located in the nucleus of the atom, and the electrons occupy the space outside the nucleus. The number of protons in the nucleus of an atom is called the atomic number, and it determines the identity of the element. For example, all atoms with 6 protons are carbon atoms, and all atoms with 8 protons are oxygen atoms.
The number of neutrons in an atom can vary, and atoms of the same element can have different numbers of neutrons. These atoms are called isotopes. The number of neutrons in an atom is not included in the atomic number, but it does affect the atomic mass (the total number of protons and neutrons in an atom).
The electrons in an atom are arranged in shells around the nucleus. The arrangement of the electrons in the outermost shell determines the chemical properties of the element. Elements with a full outer shell are generally unreactive, while elements with an incomplete outer shell are more reactive because they are seeking to gain or lose electrons to achieve a full outer shell.
The structure of an atom, including the number of protons, neutrons, and electrons it has, determines the properties of the element. For example, the atomic structure of carbon (which has 6 protons and 6 electrons in its outer shell) gives it the ability to form strong chemical bonds with other elements and make a wide variety of compounds, while the atomic structure of neon (which has 10 protons and a full outer shell of electrons) makes it unreactive and gives it a number of unique properties.
Also Read : Slogans on Save Earth
How Does the Earth’s Atmosphere Protect Life on the Planet?

The Earth’s atmosphere is a layer of gases that surrounds the planet and protects life on it in several ways. Some of the ways in which the atmosphere protects life on Earth include:
- Protection from ultraviolet radiation: The atmosphere absorbs harmful ultraviolet (UV) radiation from the sun, which can damage DNA and cause skin cancer. Without the atmosphere’s protection, life on Earth would not be able to survive the intense UV radiation from the sun.
- Temperature regulation: The atmosphere helps to regulate the temperature on Earth by trapping some of the heat from the sun, which keeps the planet warm enough for life to thrive. Without the atmosphere, the temperature on Earth would be too cold for most organisms to survive.
- Protection from meteorites: The atmosphere protects the Earth from meteorites and other debris from space by burning them up as they enter the atmosphere, which prevents them from hitting the surface of the planet.
- Air quality: The atmosphere helps to filter out harmful gases and particles from the air, which helps to maintain the air quality on Earth and protect life from the harmful effects of pollution.
Overall, the Earth’s atmosphere is essential for the survival of life on the planet, and it plays a vital role in protecting life from the harsh conditions of space.
What Are the Different Types of Energy and How Can They Be Transformed From One Form to Another?
Energy is the ability to do work or produce change, and it exists in many different forms. Some common types of energy include:
- Thermal energy: Thermal energy is the energy of heat and is produced by the movement of particles in a substance. It can be transformed from one form to another through the process of heat transfer, which occurs when thermal energy is transferred from a hotter object to a cooler object.
- Kinetic energy: Kinetic energy is the energy of motion and is possessed by an object that is moving. It can be transformed from one form to another through the process of work, which is the transfer of energy from one object to another through a force.
- Gravitational potential energy: Gravitational potential energy is the energy stored in an object due to its position in a gravitational field. It can be transformed from one form to another through the process of falling, during which the gravitational potential energy is converted into kinetic energy.
- Elastic potential energy: Elastic potential energy is the energy stored in an object due to its ability to return to its original shape after being deformed. It can be transformed from one form to another through the process of stretching or compressing an object, which converts the elastic potential energy into kinetic energy.
- Chemical energy: Chemical energy is the energy stored in the bonds between atoms in a molecule. It can be transformed from one form to another through the process of a chemical reaction, during which the chemical energy is converted into other forms of energy, such as heat or light.
Overall, energy can be transformed from one form to another through a variety of processes, and it is important to understand how these transformations occur in order to understand the behavior of energy in the world around us.
How Does the Nervous System Function and How Does It Coordinate the Body’s Responses?
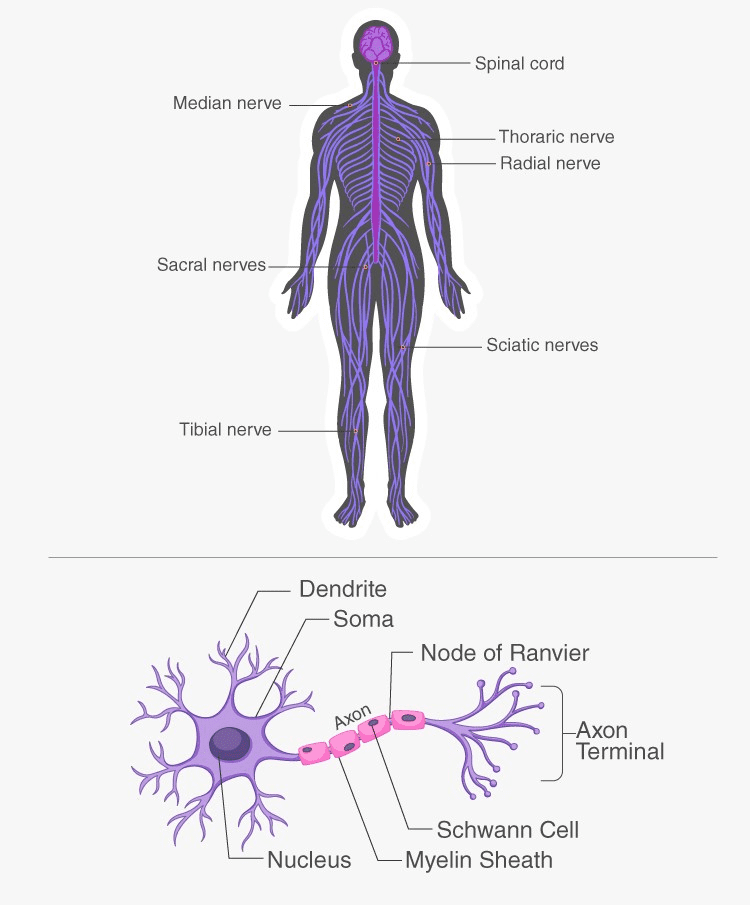
The nervous system is a complex network of cells, tissues, and organs that controls the body’s responses to internal and external stimuli. It is made up of two main components: the central nervous system (CNS) and the peripheral nervous system (PNS).
The CNS consists of the brain and the spinal cord and is responsible for receiving and processing information from the senses and coordinating the body’s responses to that information. The PNS consists of all the nerves that extend outside the CNS and carries information to and from the CNS.
The nervous system functions by transmitting and processing information through the use of nerve cells, or neurons. Neurons are specialized cells that are able to transmit electrical and chemical signals through the body. They are responsible for receiving, processing, and transmitting information to and from the CNS.
The nervous system coordinates the body’s responses to stimuli through the use of reflexes, which are rapid, automatic responses to stimuli that do not require conscious thought. Reflexes are controlled by the spinal cord and are important for maintaining homeostasis and protecting the body from harm.
In addition to reflexes, the nervous system also coordinates the body’s responses to stimuli through the use of the brain and spinal cord. The brain processes information from the senses and sends signals to the appropriate muscles, glands, or organs to coordinate the body’s responses.
Overall, the nervous system plays a vital role in the functioning of the body by controlling and coordinating the body’s responses to stimuli and maintaining homeostasis.








Recent Comments